Authors: Ghassab Al-Mazaideh, Ashraf Al-Msiedeen, Fadi Alakhras, Hammad Aldal’in,Haya Salman,Zeinab Al-Itiwi, Khaled Al Khalyfeh,Salim Khalil
DOI: https://doi.org/10.48103/jjeci142018
JORDANIAN JOURNAL OF ENGINEERING AND CHEMICAL INDUSTRIES (JJECI)
Pages: 38-68
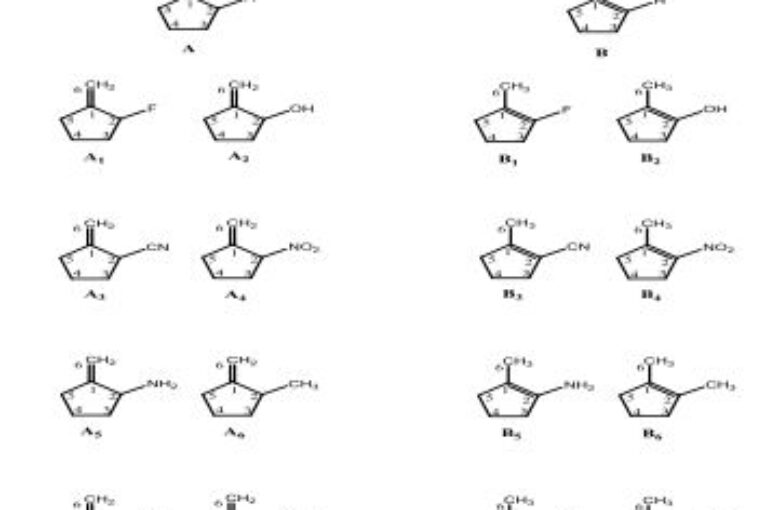
In this study, the geometry optimizations, orbital energies (HOMO-LUMO) and relative stabilities of methylene cyclopentane and 1-methylcyclopentene were investigated by DFT
calculations. 1-methylcyclopentene was found to be more stable than methylene cyclopentane isomer with enthalpy value H=18.518 kJ/mol. Also, the effect of substituents X (F, OH,
CH3, NH2, CN, NO2, CHO and CF3) also studied on the relative stabilities of these two tautomers. The results showed that the stability of both isomers is increased by all substitutes.
Gibbs free energy calculations have been used to find the effect of substituents X on the system.
Keywords: methylene cyclopentane, 1-methylcyclopentene, substituent, tautomer, DFT