Authors: Malek M. A. Al-Daradka, Omar S. Al-Ayed and Eyad S. M. Abu-Nameh
DOI: https://doi.org/10.48103/jjeci8142025
JORDANIAN JOURNAL OF ENGINEERING AND CHEMICAL INDUSTRIES (JJECI)
Pages: : 129-135

Highlights
- Oil shale crushed and pyrolyzed at 550 oC
- Shale oil is treated with ionic liquids
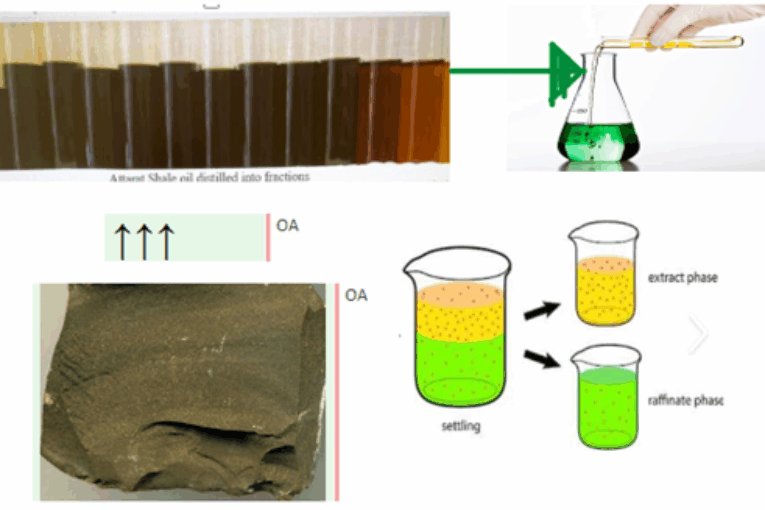
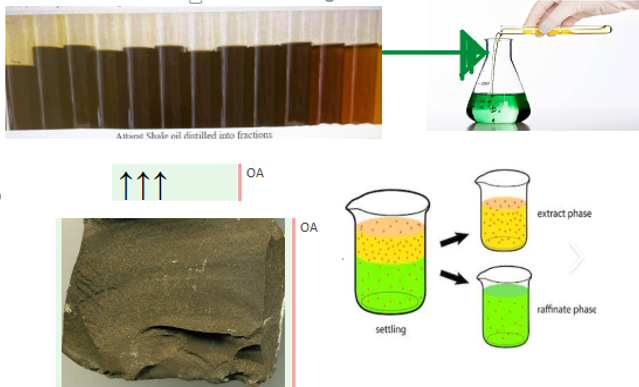
- Oil is separated into two layers
- Rich sulfur compounds and lean phase are tested for sulfur compounds
- Determination of total sulfur in each phase
Abstract
Oil shale is a fine-grained sedimentary rock that contains large quantities of organic materials, mostly kerogen. Jordan has large reserves of oil shale, estimated at more than 70 billion tons, oil shale contains sulfur in the form of pyrite compounds. Organometallic sulfurs are found in shale oil after pyrolysis of the oil shale. The sulfur could be up to 10 wt.% of the total shale oil generated. Oil Shale sample collected from Attarat Um Ghudran mine east of Amman – Aqaba desert highway. In this research, two types of ionic liquid were used to extract sulfur from shale oil: 1-ethyl-3-methylimidazolium chloride ([EMIM][Cl]) (IL-A) and 1-butyl-3-methylimidazolium thiocyanate ([BMIM][SCN]) (IL-B). Ionic liquid was mixed with shale oil in a ratio (1:1) for the extraction of sulfur compounds. Results have shown good ability of ILs to extract sulfur present in shale oil. Results indicate that the removal efficiency of IL-A ranged from 28% to 38.9%, while IL-B efficiency ranged from 30.8% to 41.4%. Ionic liquids have succeeded in extracting organometallic sulfur from shale oil.
Paper type:Research Paper
Keywords: Sulfur, Extraction, Ionic liquid, Shale Oil, Pyrolysis.
Citation: Al-Daradka, M., O., Al-Ayed , and E. Nameh ” Removal of Sulfur-Containing Compounds from Shale Oil Using Ionic Liquids, Vol. 8, No.2, pp:129-135 (2025).