Authors: Bara’ K. Al-Mahameed
DOI: https://doi.org/10.48103/jjeci942026
JORDANIAN JOURNAL OF ENGINEERING AND CHEMICAL INDUSTRIES (JJECI)
Pages: 62- 44

Abstract
Highlights
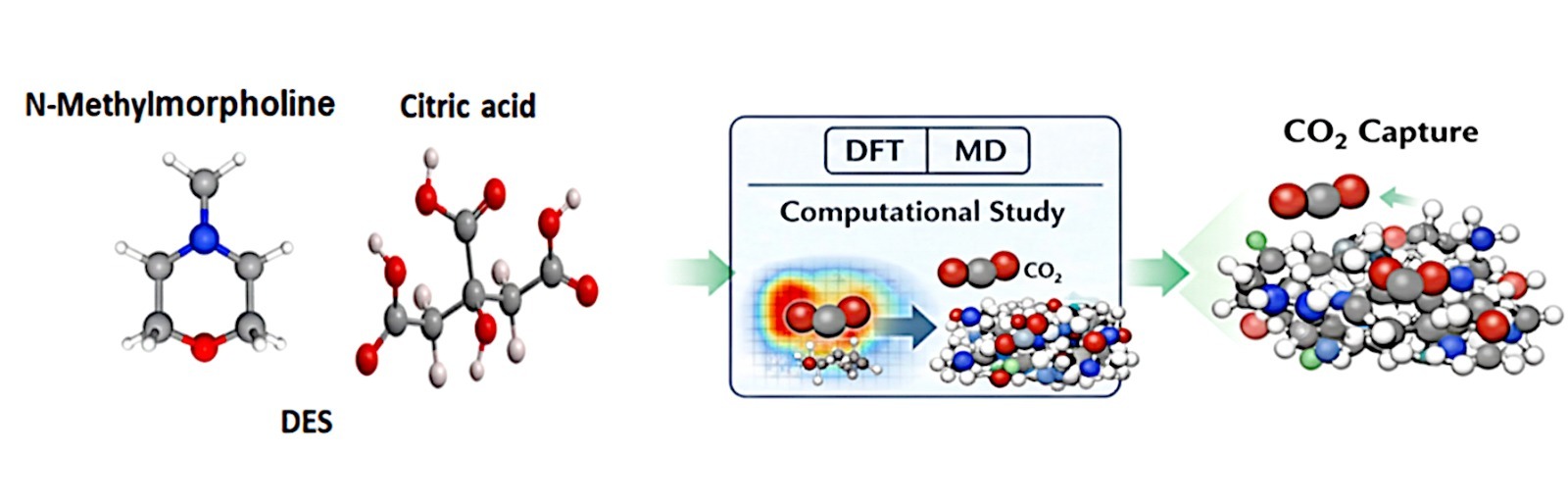
• Citric acid and N-Methylmorpholine form stable and efficient DESs.
• DESs studied as novel solvents for CO₂ capture.
• Diffusion coefficient of CO2 in DES is 4.879× 10⁻12 m2/s.
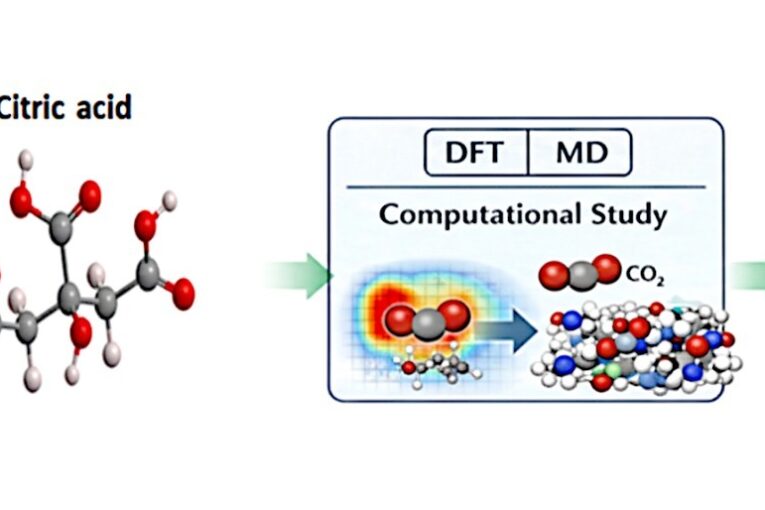
In recent years, scientists and researchers have been interested in developing effective methods to reduce atmospheric CO2 levels, due to their negative effects on the environment and human health. Deep eutectic solvents are considered promising alternatives for the future due to their attractive and distinctive properties. In this study, the citric acid and N-methylmorpholine were mixed to obtain a novel DES by forming a hydrogen bond between two components. Computational simulations, including molecular dynamics using LAMMPS with the General AMBER Force Field and density functional theory calculations, were performed to investigate CO₂ absorption behavior. The simulation system consisted of 140 citric acid molecules, 200 N-methylmorpholine molecules, and 40 CO₂ molecules. Structural,
transport, and reactivity properties were analyzed using radial distribution functions (RDF), mean square displacement (MSD), and quantum chemical descriptors. RDF analysis revealed pronounced intermolecular interactions between CO₂ molecules and the functional groups of the DES components, indicating preferential structuring around the solvent molecules. The diffusion coefficient calculated
from the MSD analysis was 4.879 × 10⁻¹² m² s⁻¹, suggesting moderate mobility of CO₂ molecules within the solvent environment.
Paper type: Research Paper
Keywords: Deep eutectic solvent, Capture of CO2, DFT, Molecular modeling, Computational simulation.
Citation: Al-Mahameed, B. K. M., “CO₂ Capture Using a Citric Acid and N-Methylmorpholine Deep Eutectic Solvent: A Computational Study,”
Jordanian Journal of Engineering and Chemical Industries, Vol. 9, No. 1, pp: 44–62 (2026).333