Authors: Niveen W. Assaf, Ibukun Oluwoye, Ali Marashdeh and Ali Elrashidi
DOI: https://doi.org/10.48103/jjeci8202025
JORDANIAN JOURNAL OF ENGINEERING AND CHEMICAL INDUSTRIES (JJECI)
Pages: : 214-226

Highlights
- Stability of water over actinide oxides was revealed.
- Dissociative adsorption is predominant on (100) and (110) surfaces.
- (111) surfaces were found to have a stable mixed water layer.
- Nanoparticles will adopt octahedral and truncated-octahedral shapes in moist media.
- The study provides insights into corrosion and catalysis.
Abstract
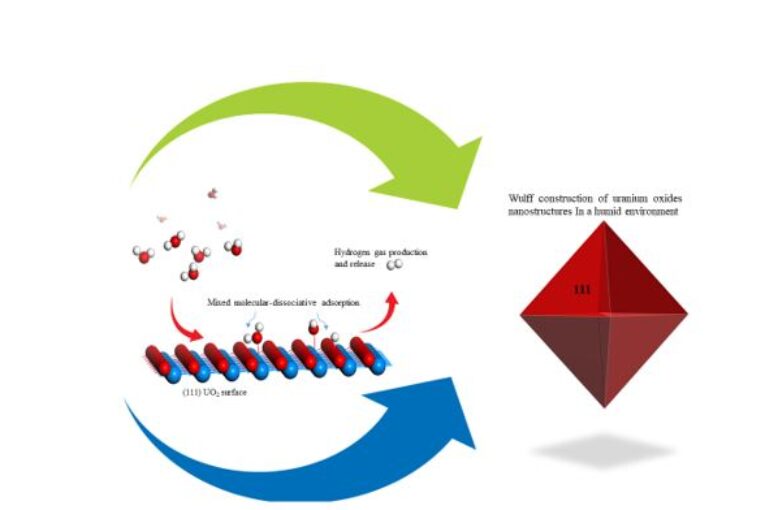
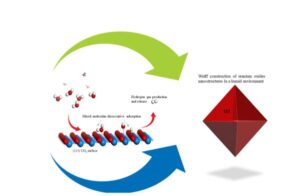
The surface free energy diagrams of water adsorption on the low-index surfaces of UO2 and PuO2 were calculated using the ab initio atomistic thermodynamics technique. The phase diagrams of both UO2(100) and PuO2(100) surfaces show that the dissociative H-OH adsorption of water exhibits higher stability for all applied coverages. Chemical potential, with PuO2(100) stabilizing hydroxylated structures at lower chemical potentials than UO2(100). On the (110) surfaces, dissociative adsorption is also preferred, but with reduced reactivity compared to (100); UO2(110) supports hydroxylation at lower chemical potentials than PuO2(110), indicating a greater tendency for early-stage protonation. On the closed-packed (111) surfaces, the stability diagrams revealed that only a mixed monolayer containing both molecular and dissociated water becomes stable, and only under more humid or elevated temperature conditions. The mixed adsorption phase on PuO2(111) becomes stable at a lower chemical potential relative to UO2(111), indicating slightly higher reactivity. The equilibrium morphology of UO2 and PuO2 nanoparticles in humid environments were predicted using the Wulff construction. The results showed that the (111) facets dominate across the full range of water chemical potentials, yielding octahedral nanoparticles. For PuO2, a transition to truncated octahedral was observed eV due to the emergence of (110) facets. These findings clarify the role of thermodynamic and kinetic variables affecting water interactions with actinide oxides, pertinent to corrosion, catalysis, and the performance of nuclear materials.
Paper type: Research Paper.
Keywords: uranium dioxide, plutonium dioxide, adsorption; DFT, wulff construction, stability, thermodynamic.
Citation: Assaf, N., W., Oluwoye, I., Marashdeh, A., and Elrashidi, A. “Atomistic thermodynamics of water over actinide oxide surfaces: implications on stabilities and morphologies” Jordanian Journal of Engineering and Chemical Industries, Vol. 8, No.2, pp:214-226 (2025).